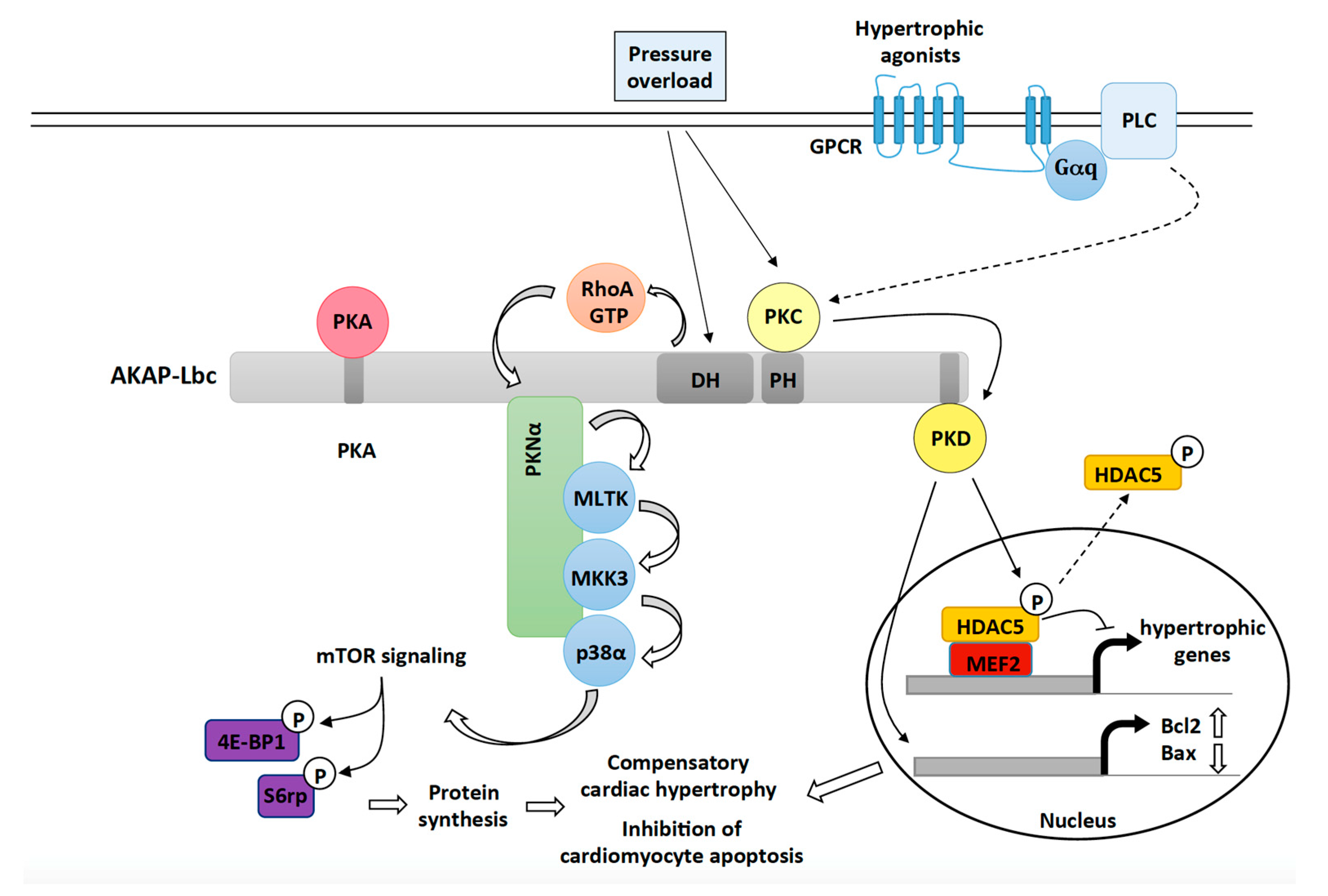
The formation of cytosolic signalling condensates is based on two processes. But how are the cytosolic proteins trafficked to their cognate membrane nanoclusters? Mounting evidence has emerged recently that a large number of membraneless compartments (also called non-membrane-bound organelles or biomacromolecular condensates) are assembled via LLPS 12. Membrane-resident signalling proteins, such as transmembrane (TM) and membrane-associated proteins, are predominantly trafficked to the plasma membrane via the secretory pathways 11. RAS nanoclusters form and locally increase the concentration of RAS paralogs in membrane microdomains 10. For example, activated protein nanoclusters in specialised membrane microdomains selectively connect with and subsequently activate cytosolic signalling components or complexes 8, 9. Rapidly emerging reports on signalling networks support the idea that various signalling molecules operate together in functional protein complexes. Structural and functional variety of accessory proteins What the functions of the accessory proteins are, why are they crucial for signal transduction, and whether they represent better therapeutic targets for different human diseases are questions that will be addressed in this article in the context of the RTK-RAS-MAPK signalling pathway. 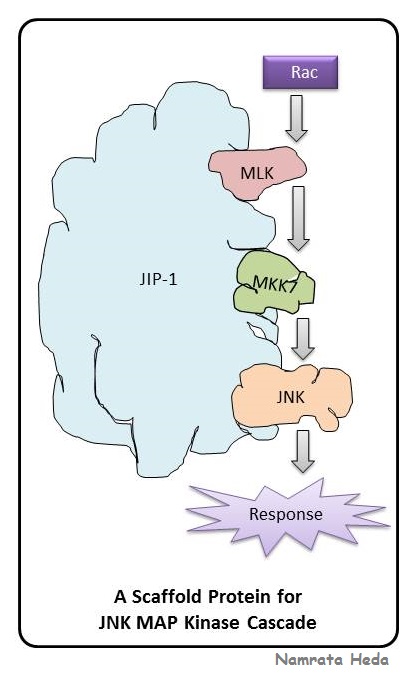
Accessory proteins link constituent components of individual signal transduction pathways by forming physical complexes. A large number of proteins, hereafter, collectively designated as the ‘accessory proteins’, fulfil the requirements to drive LLPS and have been reported to act as adaptor, anchoring, docking or scaffold proteins. This state can be achieved by liquid–liquid phase separation (LLPS), a mechanism in which two separate liquid phases with different protein compositions emerge from one mixed solution 7. However, the strength, efficiency, specificity and accuracy of signal transduction are controlled by mechanisms that increase the connectivity of the signalling molecules and thus increase their local concentration and reduce their dimensionality. This signalling cascade has rapidly taken centre stage in cancer and RASopathy therapies (see below). Dysregulation of components or regulators of this cascade is frequently associated with tumour growth and a distinct subset of developmental disorders called the RAS-MAPK syndromes or RASopathies 4, 5, 6. The RTK-RAS-MAPK axis is a highly conserved, intracellular signalling pathway that has an essential role throughout mammalian development, from embryogenesis to tissue-specific cellular homoeostasis in the adult 3. 
This pathway contains a three-tiered kinase cascade comprising the serine/threonine kinases ARAF/BRAF/CRAF, the dual specificity kinase MEK1/2 and the serine/threonine kinases ERK1/2 1, 2. Activated receptor tyrosine kinases (RTKs), for instance, link the RAS activator SOS1 to RAS paralogs, e.g., the proto-oncogene KRAS4B, which in turn regulate various signalling pathways, including the mitogen-activated protein kinase (MAPK) pathway 1. Signals are processed by evolutionarily conserved signalling cassettes that comprise specific constituent components acting as receptors, mediators, effectors and regulatory proteins. Nature has evolved sophisticated, cell type-specific mechanisms to sense, amplify and integrate diverse external signals, and ultimately generate the appropriate cellular response.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
